The Alkaline-Powered No. 6 Battery for Impulse-Wound Clocks
Ken Reindel Ken’s Clock Clinic
Imagine the ideal No. 6 vintage battery for self winding clocks:

- Outputs 3.3 volts regardless of internal battery state of charge.
- Powers any 3 volt clock from the stubborn American Clock Co. impulse wound to the troublesome Broadcast Studio models with miniature dc motors.
- Run time 2 years on two alkaline D cells.
- Efficiently uses all of the energy from the internal alkaline D cells, maximizing run time and conserving the environment.
- Very affordable.
The above describes the Model 1900G, packaged as a great-looking vintage style No. 6 unit. It will power ANY 3 volt self winding clock, without hesitation, and without detriment to the clock.
Alkaline battery technology by itself has greatly advanced. Alkaline D cells are quite incredible for what they cost. They fall short when you need high impulse energy to wind clocks like American Clock Co. or Hipp-Toggle models. Until now, we could only offer a rechargeable model (the Model 1900R) for these demanding clocks.
When you use alkaline batteries alone to power your American Clock Co. impulse style clock, or any other impulse-wound clock, there comes a point in time when the battery will not wind the clock anymore. When alkaline D batteries have been depleted of only 1/3 their capacity, their available impulse energy drops off substantially. At that point, they cannot reliably run your clock. You replace the batteries and the cycle starts all over. Unfortunately you’re throwing away good batteries with energy left in them, which is not very good for our environment. It’s the equivalent of throwing away gasoline.
The Model 1900G converts this remaining energy into usable impulse energy that runs the clock. To do this, it processes the alkaline battery output through two award-winning, breakthrough green circuit technologies. The result is output recovery capability very similar to the original (now long since obsolete) No. 6 batteries—but with much longer life. With the Model 1900G you’ll get every ounce of energy from the internal D batteries before surrendering them, fully spent, to the recycle bin.
See below for Technical Overview.
The Model 1900G is affordably available at www.kensclockclinic.com .
Model 1900G Technical Overview
We will now describe what makes the 1900G function. This might at first seem like a lot of fuss to power antique clocks. But, if we are to conserve these clocks for generations to follow, we have to do the job right. At KCC Scientific we take great pride in doing the job with exactness and precision.
We’ve extensively studied in great detail the clocks that these products are designed to power. We’ve also studied the behavior of the original No. 6 batteries and why these vintage clocks functioned so well when powered by them.
First, let’s look at the problems associated with a pair of alkaline cells driving an impulse- wound self winding clock. We have two problems to deal with:
- Output impedance of the battery very rapidly increases with use
- Terminal voltage drops as the cells are depleted.
Both of the above problems are the Achilles heels of alkaline cells. These problems often result in early visits to the recycle bin. Anyone with a high performance digital camera is familiar with this. Alkaline batteries don’t seem to last very long when used in cameras, or many other modern electronic devices, either. This problem is the same as the problems we have powering vintage impulse-driven clocks, more or less.
The early impulse-driven clocks are all alike in the following way. The clock unwinds. A switch closes. A coil of around 6 ohms is slammed across the battery. An electromagnetic field is generated by the flow of current from the battery to the coil. The clock winds, the switch opens, and the clock runs down again for the cycle to repeat.
The early impulse clocks were designed around the original No. 6 battery. These early batteries had a huge amount of plate surface area: the negative terminal was the can of the battery! That’s a lot of surface area to store charge for that once per 7 minute impulse that drove the American Clock Co., Imperial, Gregory, numerous Hipp-toggles, and other clocks of the era.
When it came time to wind, that charge was ready to be transferred to the coil, converted to plentiful electromagnetic energy, sufficient to wind the clock with authority. Today’s D cell, while packed with plentiful energy density, just doesn’t have the charge storage to do the job. So when the clock requires an impulse of energy, the battery voltage sags, and its power simply cannot be transferred to the clock coil.
Digital camera manufacturers recommend Lithium batteries to improve on this situation. These cells do a better job than alkalines, have flatter discharge curves, and better impulse energy. We’ve thought about offering a lithium battery solution. The problem is that lithiums are anywhere from 3x to 5x more expensive than alkalines. Further, 1.5 volt lithiums are only available in AA size. D size lithiums are available as 3 volt cells but are outrageously expensive and cannot output the currents needed to wind clocks.
We could of course recommend using lots of alkaline D batteries in a series-parallel combination. Some enthusiasts do this. The problem is, you’ll be throwing away that many times the number of good batteries when they do eventually droop down too low to power the clock.
By the way, we ship all of our alkaline-powered products with Energizer cells pre-installed (was Eveready, prior to that National Carbon Company). We like a couple things about Energizer. First, they are made in USA and they work very well. Second, the history— and invention–of the No. 6 Columbia Dry Battery passes through Lakewood, Ohio and the Energizer heritage. We also like that they offer good value.
We now submit for discussion the discharge curves for a typical alkaline battery.
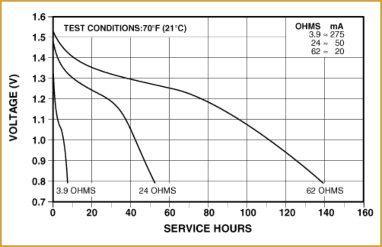
Navigating the above curve technically may seem complicated but it isn’t. Look at the curve marked “62 ohms” and forget the others for the moment. This curve shows quite simply that the voltage at the battery terminals starts out at 1.5 volts at 0 service hours. As time goes on, the voltage droops dramatically downward. Eventually the battery terminal voltage shows 1.3 volts, at about 1/3rd the service life. A pair of cells in series would show a terminal voltage of 2.6 volts at this point. At this voltage, most impulse-wound self winding clocks would probably still run, if discharge curve was the only consideration.
The other consideration is the cell’s output resistance. The manufacturer specifies around 200 milliohms fresh—each cell. Put two in series to obtain 3 volts and now we have conservatively 400 milliohms total resistance when the cells are fresh. That means that when the clock connects the 6 ohm coil to the batteries we are going to drop up to 0.2 volts across this cell resistance when the battery is fresh. Most likely you’ll get by as long as the batteries are fresh.
The problems occur when the cells have been used for a few months in the clock and these above effects start to play together. After a few months, the cell output resistance nearly doubles. Now, the voltage drop internal to the cells in response to the clock’s coil is almost 0.4 volts (assuming the resistance has doubled). This, subtracted from the 2.6 volts
we have at 1/3rd the service life, results in a 2.3 volt output, which is not likely to trigger a winding, and the clock will stop. While somewhat oversimplified, this is the nature of the problem we face using alkaline cells to drive impulse-wound clocks.
At this point, 1/3rd or so into the service life of fresh batteries, you’ll likely remove them from the clock and throw them away, thinking they are dead! Yet most of the cell life remains, if we can find a way to extract it from the battery.
This problem is not limited to powering antique clocks. In our neighborhood there is a collection depot for recycling old batteries. We went in and tested 10 alkaline cells brought in for recycling with a portable digital multimeter. Out of the 10, there were 8 that measured terminal voltages of 1.5 volts or more! According to the manufacturer, only 10% of the cell’s life has been used at this point. Most likely someone used the batteries in an electronic device requiring impulse energy. They “ran down” and the 90% good cells were discarded.
Alkaline cell manufacturers recognize this behavior. You can check out page 8 of this link if you are interested. http://data.energizer.com/PDFs/alkaline_appman.pdf This is the Energizer Applications Manual describing the technical behavior of their alkaline cells. Look at the figure on page 8, which is an excellent graphical description of what happens to the cell’s internal resistance—and resulting terminal voltage under load—as the battery capacity is used up. It is quite dramatic.
Some enthusiasts make up for the alkaline shortfall by using higher voltage, often the output of a Lantern Battery (6 volts). This works, for awhile. But eventually the higher battery voltage catches up with you. Contacts die an early death, or carbonize and corrode which requires frequent cleaning. In the meantime, the extra “jolt” from the over voltage on these 3 volt clocks wears out the mechanical components. Are we conserving the clocks this way? We submit, the answer is NO. We will NOT warranty any 3 volt clock we restore powered by 6 volts, ever.

The 1900G. There are two key “green” technologies that work together in the 1900G. First, we take the battery output from the alkalines and “condition” it in such a way that this “conditioned” output is always 3.3 volts. This is called “Boost
Regulation.” As the alkaline battery voltage droops, our circuit converts its output voltage to 3.3 volts. It regulates this voltage so that no matter what the battery voltage is, the output will always be
3.3 volts. Well, within limits. For the remarkable Texas Instruments chip used in the Model 1900G, the limit on its input (from the battery) is about 1 volt total in our 1900G, or 0.5 volts per D cell. If you’ve been around the electronics industry as long as we have, you’d realize that this is truly breakthrough. This TI part won the ECN Readers Choice Award in December 2007.
Let’s go back to the battery curve above. The theory of boost conversion does not escape conservation of energy. If we boost the battery voltage, it follows that (assuming a constant load on the 1900G output terminals) as the alkaline battery output voltage droops, the current being drawn from the batteries must increase. This makes the battery voltage
droop even more, and the boost regulator has to work harder. When the batteries are fresh, this is no problem. Ultimately there will come a point in the battery’s life where the converter will not be able to keep its output voltage constant anymore.
Enter the second “green” technology we’ve employed—Ultracapacitor technology. By using the boost converter to charge an Ultracapacitor to the proper voltage (3.3 volts in this case), we now have the energy reserve for that point in time when the alkaline cells just can’t put out enough impulse power to keep the boost converter fed. The nice thing about devices meant for clock winding applications (like the 1900G) is that clocks need a lot of energy, but for only a very short time! So our Ultracapacitor technology is perfect in tandem with the boost converter for powering impulse-wound clocks.
How large are the Ultracapacitors we use? The one we now use is 5 Farads. Yes, Farads, not microfarads. They’re called Ultracapacitors because they offer HUGE capacitance in a very small package. Output resistance is incredibly low (less than 100 milliohms, or much less than 1/2 that of two fresh alkaline cells in series). A capacitor of 5 farads mathematically allows us to output
0.5 amps for 0.2 seconds with a voltage droop of less than 0.1 volts, accounting also for series resistance of the ultracapacitors. This is more than long enough to wind an impulse clock, and there is plenty of energy reserve to get the job done thanks to the 3.3 volt boost regulator which charged that capacitor to start with. The terminal voltage never drops below 3.1 volts and positive winding is assured. And incredibly, this holds true even when the batteries are near full depletion.
The result of all this lengthy technical jargon is a No. 6 battery replacement that acts more like (actually better than) the original No. 6 battery than anything else available today. The ultracapacitor mimics the large plate area of the original No. 6 cells. The boost converter technology assures the ultracapacitor always has 3.3 volts at its terminals, so that the clock has what it needs to wind, every hour, minute, 5 minutes, or whatever the winding cycle is on the clock. In the jargon of its day, this would have been called excellent “battery recuperation.”
![]()

By the way, why do we call these technologies “green” technologies? “Green” infers extracting every ounce of available energy from an energy source with minimal waste (inefficiency). For example, the Texas Instruments chip is even capable of extracting energy from solar cells under cloudy skies, to deliver useful energy. Ultracapacitor technology has great hopes for powering electric cars in the future. This is because the power lost by charging them is a fraction of what is lost during the process of recharging batteries. These are great “green” technologies to watch as they become a substantial part of our future.